
Reusing Sulfur-Poisoned Palladium Waste as a Highly Active, Nonradical Fenton-like Catalyst for Selective Degradation of Phenolic Pollutants | Environmental Science & Technology

TEM images of reused palladium nano-spheres (a and b, after first run;... | Download Scientific Diagram

Facile Hydrogenative Deprotection of N-Benzyl Groups Using a Mixed Catalyst of Palladium and Niobic Acid-on-Carbon | ACS Omega
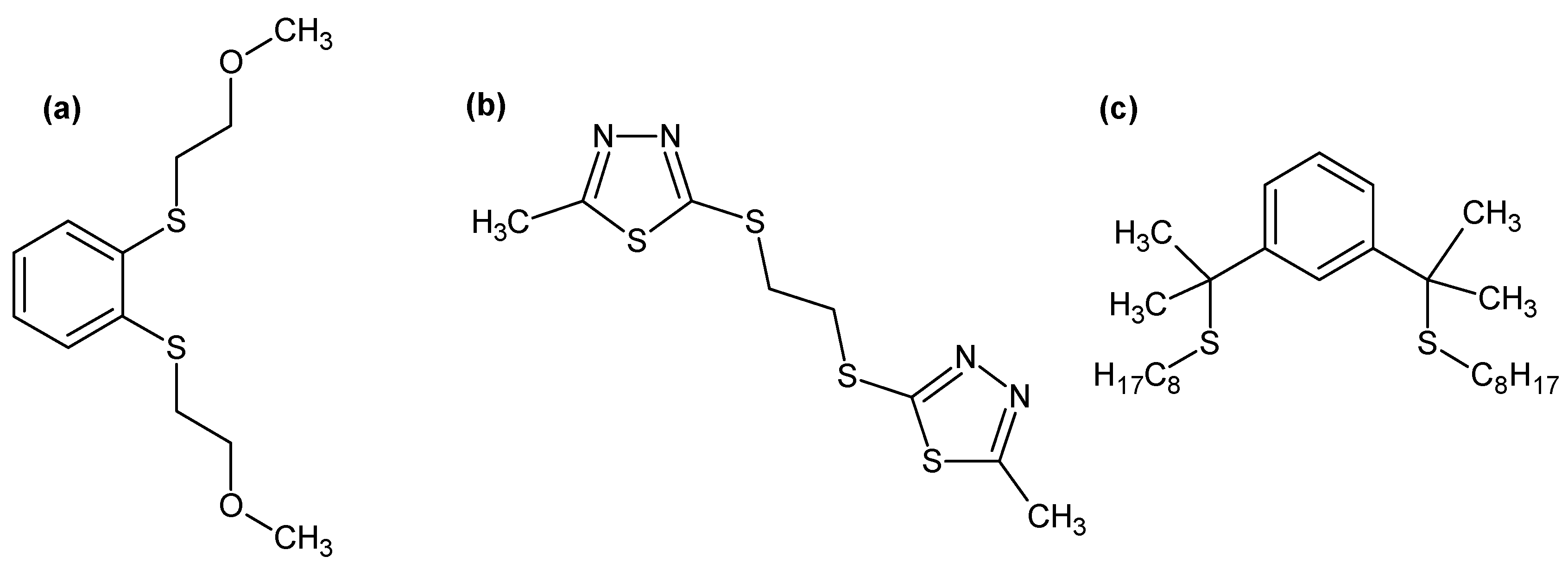
Metals | Free Full-Text | Recycling of Palladium from Spent Catalysts Using Solvent Extraction—Some Critical Points
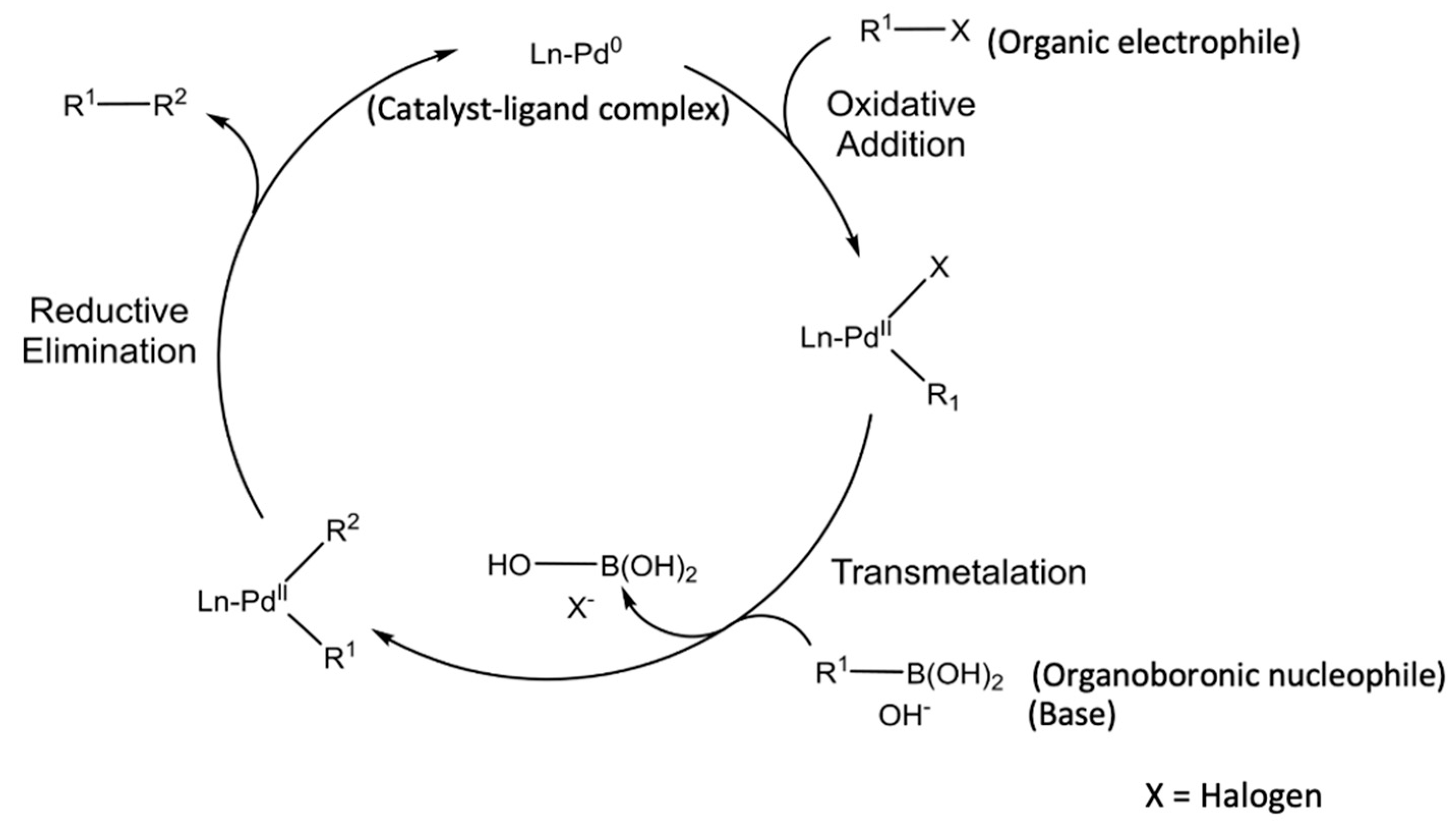
Knowledge | Free Full-Text | Catalyst Recycling in the Suzuki Coupling Reaction: Toward a Greener Synthesis in the Pharmaceutical Industry

C(sp3)−H Arylation Promoted by a Heterogeneous Palladium‐N‐Heterocyclic Carbene Complex in Batch and Continuous Flow - Ferlin - 2022 - ChemSusChem - Wiley Online Library
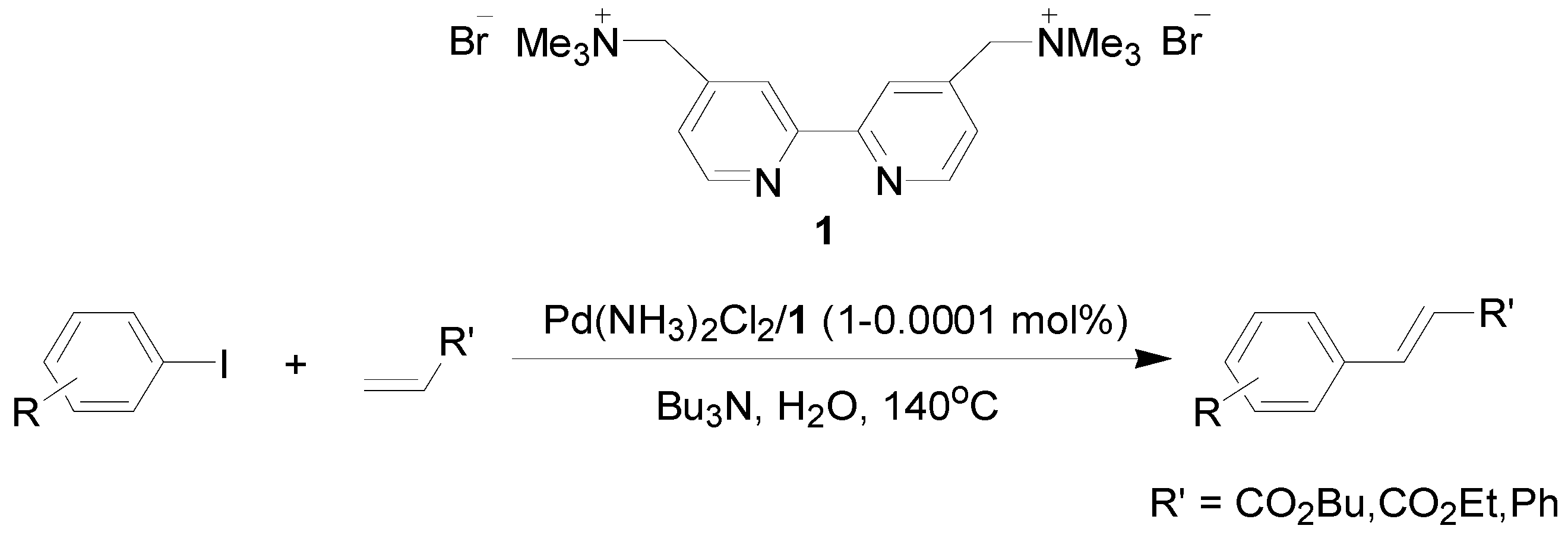
Molecules | Free Full-Text | Palladium(II)/Cationic 2,2'-Bipyridyl System as a Highly Efficient and Reusable Catalyst for the Mizoroki-Heck Reaction in Water

Selective transfer hydrogenation of phenol to cyclohexanone on supported palladium catalyst using potassium formate as hydrogen source under open atmosphere - ScienceDirect

Reusing Sulfur-Poisoned Palladium Waste as a Highly Active, Nonradical Fenton-like Catalyst for Selective Degradation of Phenolic Pollutants | Environmental Science & Technology
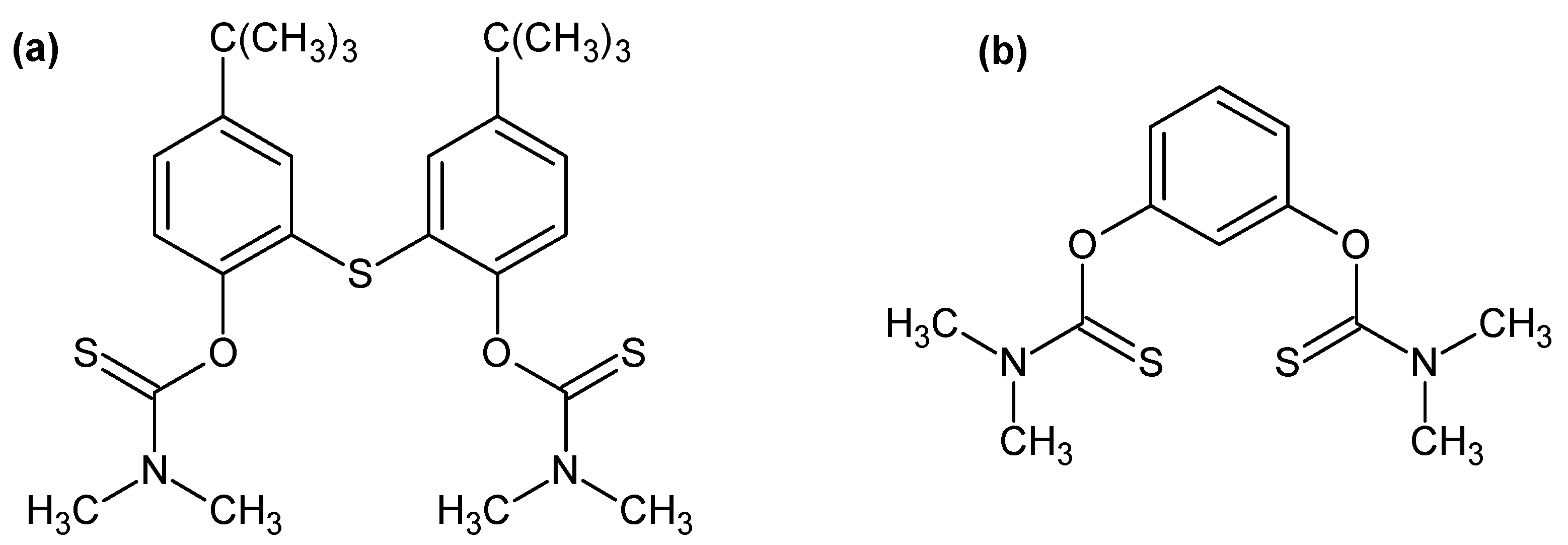
Metals | Free Full-Text | Recycling of Palladium from Spent Catalysts Using Solvent Extraction—Some Critical Points
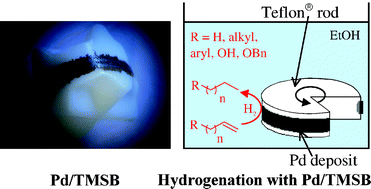
Hydrogenation versus hydrogenolysis with a safe, selective and reusable catalyst: palladium black on Teflon® - New Journal of Chemistry (RSC Publishing)

Development of reusable palladium catalysts supported on hydrogen titanate nanotubes for the Heck reaction - ScienceDirect
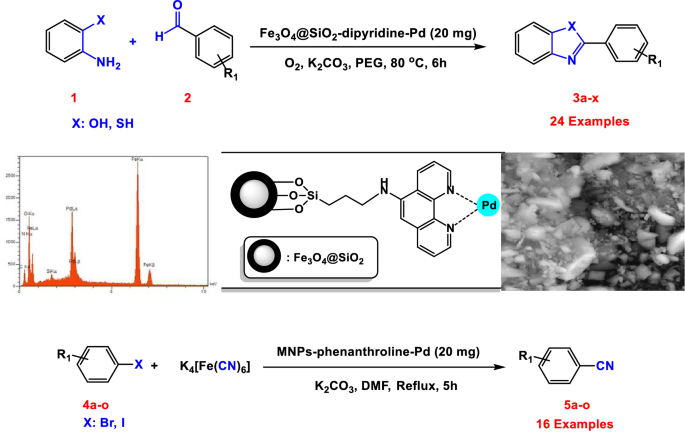
Palladium (II) Complex Supported on Magnetic Nanoparticles Modified with Phenanthroline: A Highly Active Reusable Nanocatalyst for the Synthesis of Benzoxazoles, Benzothiazoles and Cyanation of Aryl Halides | SpringerLink

Palladium supported on poly (ionic liquid) entrapped magnetic nanoparticles as a highly efficient and reusable catalyst for the solvent-free Heck reaction - ScienceDirect










